|
This article needs additional citations for. Unsourced material may be challenged and removed. (April 2009) () Arrow pushing or electron pushing is a technique used to describe the progression of mechanisms. It was first developed. In using arrow pushing, 'curved arrows' or 'curly arrows' are superimposed over the of reactants in a to show the. 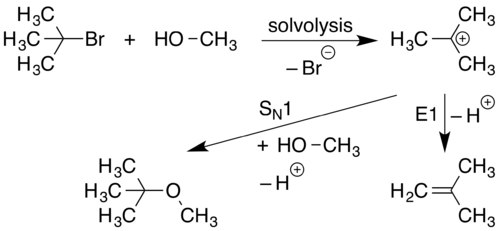
The arrows illustrate the movement of as between are broken and formed. Arrow pushing is also used to describe how positive and negative are distributed around through. It is important to remember, however, that arrow pushing is a formalism and electrons (or rather, electron density) do not move around so neatly and discretely in reality. Pushing electrons by daniel p weeks pdf - WordPress.com. Pushing Electrons 4th Edition by Weeks, Daniel P. Textbook PDF Download archived file. Daniel Hannan Telegraph Blogs Daniel Hannan Daniel Hannan is the. Because we will declare the package around 60USD and declare the package as DVD player. Very few packages may be delayed by customs verification processes that are different in each country.Insurance will be included in the shipping. However,if import duty is required, it should be paid by customers. Import duty is free in most countries for most cases. Where to vietnamese karaoke songs for. Please provide correct shipping address and telephone number when you place the order. Recently, arrow pushing has been extended to, especially to the chemistry of s- and p- elements. It has been shown to work well for compounds. Trajectory of single electron When a bond is broken, electrons leave where the bond was and this is represented by a curved arrow pointing away from the bond and ending the arrow pointing towards the next unoccupied molecular orbital. Similarly, organic chemists represent the formation of a bond by a curved arrow pointing between two species. For clarity, when pushing arrows, it is best to draw the arrows starting from a lone pair of electrons or a σ or π bond and ending in a position that can accept a pair of electrons, allowing the reader to know exactly which electrons are moving and where they are ending. Bond are broken in places where a corresponding antibonding orbital is filled. Some authorities allow the simplification that an arrow can originate at a formal negative charge that corresponds to a lone pair. However, not all formal negative charges correspond to the presence of a lone pair (e.g., the B in F 4B -), and care needs to be taken with this usage. Breaking of bonds [ ] A joining atoms in an organic molecule consists of a group of two electrons. Such a group is referred to as an electron pair. Reactions in organic chemistry proceed through the sequential breaking and formation of such bonds. Organic chemists recognize two processes for the breaking of a chemical bond. These processes are known as homolytic cleavage and heterolytic cleavage. Homolytic bond cleavage [ ] Homolytic is a process where the electron pair comprising a bond is split, causing the bond to break. This is denoted by two single barbed curved arrows pointing away from the bond. The consequence of this process is the retention of a single unpaired electron on each of the atoms that were formerly joined by a bond. These single electron species are known as. For example, light causes the -chlorine bond to break homolytically. This is the initiation stage of. Heterolytic bond cleavage [ ] Heterolytic bond cleavage is a process where the electron pair that comprised a bond moves to one of the atoms that was formerly joined by a bond. The bond breaks, forming a negatively charged (an ) and a positively charged species (a ). The anion is the species that retains the electrons from the bond while the cation is stripped of the electrons from the bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
